
How it started
Spectroscopes play an important role in modern analytical science, with applications in very different areas such as forensics, quality control and astronomy. The determination of presence and concentration of the elements in a sample is an important part of the work of modern analytical laboratories. But the spectroscope also played a crucial role in the development of modern science.
One of the first objects to be spectroscopically studied was the sun, and at the beginning of the 19th century it was first established that the rainbow spectrum disclosed by Isaac Newton was not continuous. The dark stripes, which we now refer to as absorption lines, were initially called Fraunhofer lines. After the German scientist who systematically mapped them with his new invention – the spectroscope. His studies led to the discovery that each element possessed a characteristic line or a series of lines, allowing it to be uniquely identified.
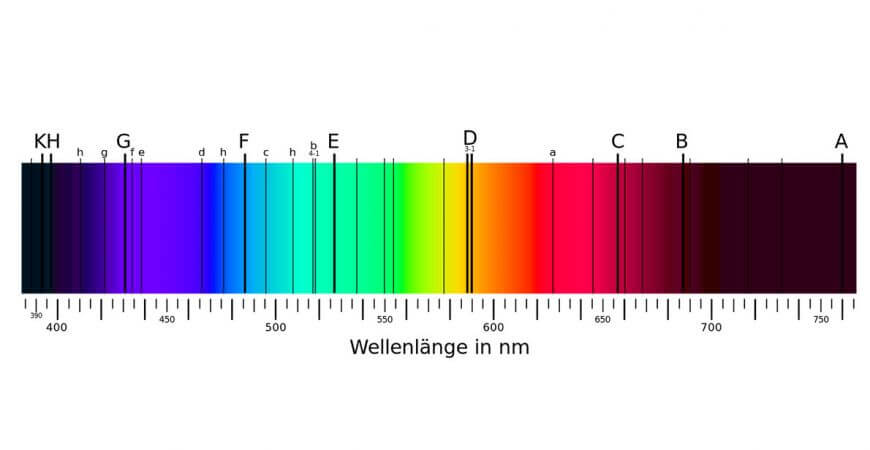
Graphical representation of the Fraunhofer lines
The work of many scientists in that century led to the progressive development of the spectra of numerous elements, but a mystery represented the spectral line of the sun at 587.49 nm, which could not be attributed to any known element. The astronomer Norman Lockyer assumed that this must be an element that was previously unknown on Earth; it was named helium – after the Greek word for the sun, Helios. Today we know that helium is the second most abundant element in the universe, but its existence was unknown before a spectroscope decoded it.
Joseph von Fraunhofer

Fraunhofer (1787-1826) examined the sunlight with a prism spectroscope, observing dark lines in the spectrum of light. In 1820, the English chemist Wollaston already made this observation. They got their name Fraunhofer lines because Fraunhofer examined them more intensively and systematically cataloged them. From 1815 he recorded about 500 spectral lines, where he first marked the lines with letters. The most striking lines are still referred to today, such as the D lines of sodium or the H and K lines of calcium.
Kirchhoff and Bunsen

Gustav Robert Kirchhoff (1824-1887) and Robert Wilhelm Bunsen (1811-1899) developed a spectroscope that was more accurate than the prism spectroscope used by Fraunhofer.

In the Kirchhoff-Bunsen spectroscope, light passes through an adjustable slit through a lens onto a prism. After the wave components of the light are refracted differently at the prism, they enter an adjustable observation tube, in which a sharp image is produced by lenses and eyepiece. The wavelength is then read on a scale that is projected by a scale tube in the beam path.
They were able to show that the black D-line in the solar spectrum at about 589 nanometers agrees with the line produced by sodium salts in the flame. Thus, both scientists developed the first spectral-analytical method and discovered the hitherto unknown elements rubidium and cesium.
From the flame test to the atomic absorption spectrometer (AAS)
Kichhoff and Bunsen were able to show that a specific spectral line belongs to each chemical element. What was new was that the specific spectral lines were completely independent of the chemical compound. This was the decisive basis for the development of AAS as an elementary analytical process. After decades of expanding theoretical foundations, Max Planck discovered the quantum absorption and emission of radiation and realized that every atom can only absorb and emit radiation of a specific wavelength.
In the mid-nineteenth century, Walsh and Alkemade independently published the analytical methods of atomic absorption spectrometry.
Worth reading about spectroscopy in the World Wide Web


Comfortably warm light in the bitter cold
A PDF article “Museumsstücke” of the observatory-museum of the University of Vienna on the history of spectroscopy.
to the contribution PDF
Seeing the stars with spectroscopy
02 March 2015: Interview with Dr Josh Nall, Whipple Museum.
Part of the show Eureka Streaker: Experiments that Changed the World